Introduction
The vagus nerve is one of the most important nerves in the human body. It connects the brain to several major organs and plays a key role in regulating automatic bodily functions. Far from being a modern wellness buzzword or a quick-fix shortcut to relaxation, this vast cranial highway serves as the main command line for your body’s unconscious processing systems. Every second you are alive, this nerve silently transmits crucial operational data that keeps your heart pumping, your lungs expanding, and your internal organs working in perfect harmony.
It is part of the nervous system that works without conscious control, helping the body maintain internal balance. Because it operates beneath your conscious awareness, your daily environments, stress burdens, and routines directly shape the quality of the signals it carries. You can explore more from trusted sources such as Britannica and Cleveland Clinic.
During my 6+ years working within clinical public health systems and primary health centres, including NRHM facilities, evaluating autonomic exhaustion and counseling individuals dealing with chronic stress-induced illness was a routine operational requirement. In these busy community clinics, we frequently observed patients presenting with unexplained digestive issues, elevated blood pressure, and severe physical burnout. They were often searching for complex medical answers, completely unaware that their high-stress lifestyles had trapped their nervous systems in a state of permanent survival alert, leaving little room for natural recovery.
My public health background taught me that you cannot force a stressed, hyper-reactive nervous system into a state of authentic calm overnight. Your body’s autonomic balance relies on steady, predictable daily habits rather than short-term trends. Reclaiming your physical equilibrium requires building a dependable lifestyle routine that protects your neural pathways and supports your body’s natural rest cycles from the ground up.
What Is the Vagus Nerve?
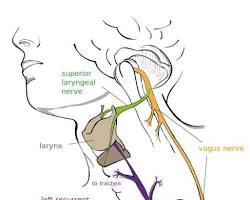
The vagus nerve (also called cranial nerve X) is the longest cranial nerve in the body. The word vagus translates to “wanderer” in Latin, an incredibly accurate name given the way this extensive neural network weaves throughout your upper body.
The nerve follows a highly complex path through your anatomy:
- It runs directly from the brainstem down through the neck.
- It travels deep into the chest cavity.
- It extends all the way into the abdomen.
Because of its wide reach, it connects the brain with many internal organs. This extensive network acts like an internal biological superhighway, allowing your brain to monitor and adjust the physical state of your organs in real time.
What Does the Vagus Nerve Do?
The vagus nerve helps regulate several important involuntary functions that are absolutely vital for survival. It operates continuously in the background, carefully matching your organ functions with the shifting demands of your daily life.
These essential involuntary tasks include:
- Heart rate: Modulating your heart rhythm to ensure efficient blood delivery based on your activity level.
- Breathing patterns: Influencing your lung function and helping coordinate smooth respiration rhythms.
- Digestion: Triggering the release of digestive enzymes and managing stomach contractions.
- Blood pressure regulation: Helping adjust blood vessel tone to keep your circulation stable.
- Throat and voice function: Controlling the muscles used for speaking and keeping your airway clear.
- Communication between the brain and organs: Serving as a dedicated two-way feedback loop for internal health data.
It is a key part of the parasympathetic nervous system, often described as the system that supports rest and recovery functions. When your brain senses that you are safe and secure, this pathway activates to lower your heart rate, kickstart digestion, and channel energy toward cellular repair.
📊 The Autonomic Feedback Loop
To understand how the vagus nerve functions as a bi-directional communication highway—constantly sending reports from your organs up to your brain and delivering operational commands back down—review this operational flowchart:
THE VAGUS NERVE SIGNALING AXIS
[Brainstem / Cranial Origin]
│
▲ │ │
│ ▼ │
[AFFERENT FEEDBACK PATH] [EFFERENT COMMAND PATH]
(80% of Total Fibers) (20% of Total Fibers)
│ │
│ ▼
──► Reports Tissue Inflammation ──► Lowers Sinoatrial Node Pace
──► Measures Nutrient Availability (Decreases Elevated Heart Rate)
──► Monitors Intestinal Stretch ──► Triggers Gastric Acid Release
│ │
└─────────────────────────┬─────────────────────────┘
│
▼
[Systemic Visceral Homeostasis]
(Rest, Digestion, & Physical Repair)
How the Vagus Nerve Works
The vagus nerve carries signals in both directions, making it a true conversation between your mind and your body rather than a one-way street:
- From the brain to the organs.
- From the organs back to the brain.
Interestingly, roughly 80% of the nerve fibers are dedicated to sending data up from your organs to your brain. This means your brain is constantly reading the physical state of your gut, heart, and lungs, using this real-time data to shape your mood, stress levels, and emotional clarity.
This continuous, two-way communication helps the body respond to internal conditions and maintain stability. For example:
- It can help regulate heart rhythm, stepping in to slow things down after a sudden fright.
- It supports digestion by influencing stomach and intestinal movement, ensuring food moves smoothly through your GI tract.
- It helps coordinate reflexes like swallowing and coughing, protecting your airway from accidental blockages.
Where Is the Vagus Nerve Located?
The vagus nerve begins deep within the brainstem, right at the base of your skull. From this origin point, a pair of nerves emerges—one for each side of your body—and travels downward through several major areas:
- The neck: Running down alongside the carotid artery, where it sends branches to your throat and voice box.
- The chest: Passing through the upper torso to interact closely with major blood vessels.
- The lungs and heart: Weaving into these organs to monitor your breathing and keep your heart rate balanced.
- The digestive system: Traveling through the diaphragm to spread across the stomach, liver, pancreas, and intestines.
It branches out to multiple organs along the way, making it one of the most widely distributed nerves in the body. This extensive structural path highlights why changes in your breathing or mental stress can instantly echo across your digestive system and heart rhythm.
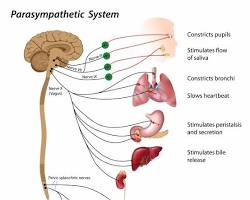
Why the Vagus Nerve Is Important
Because it connects to so many vital organs, the vagus nerve plays a major role in keeping your entire body functioning smoothly day after day. It acts as a primary buffer against the wear and tear of daily life, shielding your tissues from the damaging effects of unmanaged adrenaline and cortisol.
Specifically, this nerve is central to:
- Maintaining internal balance (homeostasis): Keeping your blood pressure, temperature, and pH levels within a safe, stable range.
- Supporting digestive function: Allowing your body to break down nutrients efficiently and maintain a healthy gut barrier.
- Regulating heart and lung activity: Making sure your heart rate adapts smoothly when you transition from exercise back to rest.
- Helping the body recover after stress: Easing your nervous system out of an alert fight-or-flight state once a challenge has passed.
It is considered a central part of the autonomic nervous system. When your vagus nerve function is resilient, your body can transition smoothly between high-energy tasks and deep, restorative recovery periods.
Understanding Integrated Metabolic Connections
Your nervous system health does not operate in a vacuum; it is deeply intertwined with your body’s broader metabolic networks, hormonal balance, and daily habits. When your lifestyle choices support your biological rhythms, your autonomic nervous system can function with minimal friction.
For instance, blood sugar stability directly influences your nerve health. If your diet is full of highly processed foods that cause sharp glucose spikes and sudden crashes, it can create an inflammatory environment that irritates delicate nerve pathways over time, disrupting smooth communication between your brain and gut.
Similarly, a chronic stress response acts as a direct roadblock to healthy vagus nerve function. When constant worries keep your sympathetic nervous system locked in a fight-or-flight state, your body continuously suppresses your rest-and-digest pathways. Giving yourself regular opportunities to relax and unwind lowers these alert signals, protecting your nerve pathways from being worn down by constant pressure.
Vagus Nerve Stimulation (Medical Context)
In medical settings, vagus nerve stimulation (VNS) refers to a highly specific therapy that uses calibrated electrical impulses to influence nerve activity. This approach is distinctly different from any casual at-home wellness practice.
It has been studied in certain clinical conditions under professional supervision, using surgically implanted devices or approved transcutaneous tools to manage complex neurological challenges. However, this type of treatment is strictly medical in nature and is only used under strict healthcare guidance. Attempting to use unverified, over-the-counter gadgets to shock or stimulate your nerve pathways without a doctor’s oversight can introduce unnecessary risks to your heart rhythm and blood pressure.
Lifestyle and Nervous System Health
While the vagus nerve cannot be directly “controlled” voluntarily, general lifestyle factors support overall nervous system balance. Your body responds best to steady, health-supporting signals that let your brain know it is safe to down-regulate and recover.
Key foundational practices that support your nervous system include:
- Regular sleep routines: Sticking to consistent sleep and wake times to give your brain a chance to clear away metabolic waste.
- Physical activity: Engaging in moderate daily exercise to build cardiovascular resilience and lower chronic stress hormones.
- Stress management practices: Incorporating habits like slow, deep diaphragmatic breathing to naturally support your body’s relaxation pathways.
- Balanced nutrition: Prioritizing whole foods packed with essential nutrients to keep your gut lining healthy and support smooth nerve communication.
- Social interaction and relaxation: Spending meaningful time with loved ones and taking regular breaks to help ease your mind out of a defensive state.
These simple, everyday habits support overall nervous system function and well-being, providing your body with the long-term stability it needs to thrive.
You may also explore:
Common Misunderstandings
Online health forums frequently oversimplify or misinterpret the science of autonomic wellness. Here are some critical, research-backed clarifications to keep your approach grounded:
- “The vagus nerve can be easily flipped like a switch” $\rightarrow$ Not true: It is part of a highly complex, interconnected nervous system that continuously balances multiple inputs. There is no single exercise or quick trick that can instantly override your body’s deeply wired survival responses.
- “Exaggerated online claims represent settled science” $\rightarrow$ In reality: Many popular internet claims are highly oversimplified versions of early laboratory research. Our scientific understanding of this nerve pathway is still developing, and there are no magical short-term fixes.
Focusing on everyday lifestyle habits is a far more reliable way to care for your nervous system than chasing viral trends that lack solid clinical backing.
When to Seek Medical Advice
Occasional feelings of mild stress or brief digestive changes are a normal part of life, often tracking with temporary project deadlines or dietary shifts. In most cases, returning to a consistent routine of rest, hydration, and gentle movement is enough to restore your body’s natural balance.
However, if someone experiences persistent or severe symptoms, it is important to consult a qualified healthcare professional for proper evaluation. You should always seek clinical guidance if you experience:
- Frequent fainting or sudden dizzy spells.
- An irregular or persistently racing heart rate.
- Difficulty swallowing or a chronic feeling of throat tightness.
- Ongoing, painful digestive issues that do not clear up with basic dietary adjustments.
Persistent, unchanging issues can sometimes point to underlying health conditions that require a formal medical assessment rather than simple lifestyle updates.
Final Thoughts
The vagus nerve is a vital part of the body’s communication system between the brain and internal organs. It plays a central role in maintaining automatic bodily functions such as heart rate, digestion, and breathing, serving as a biological anchor for your daily physical recovery.
Understanding it in a simple, scientific way helps avoid confusion created by exaggerated online claims. True, long-term well-being is not about searching for a perfect lifestyle hack, but rather about creating a steady, supportive daily routine. By protecting your sleep structure, moving your body daily, and managing your stress levels, you give your nervous system the perfect environment to stay resilient, balanced, and healthy for years to come.
Frequently Asked Questions (FAQ)
1. How does deep, slow breathing interact with vagus nerve signaling?
When you take slow, prolonged exhalations, it increases pressure within your chest cavity. This pressure change triggers specialized sensors in your blood vessels to signal your brainstem via the vagus nerve, which in turn prompts your heart to lower its pace, helping soothe your nervous system.
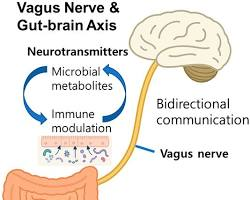
2. What role does the vagus nerve play in the “gut-brain connection”?
The vagus nerve acts as the primary physical bridge for the gut-brain connection. Specialized cells in your digestive tract monitor gut inflammation and nutrient levels, sending real-time electrical reports directly up the nerve to your brain, which can influence your mood and daily energy levels.
3. Why can chronic, unmanaged mental stress cause digestive issues?
When you are under constant stress, your sympathetic nervous system stays locked in a fight-or-flight state, which actively dampens your vagus nerve pathways. This drop in parasympathetic signaling slows down stomach contractions and reduces digestive fluids, frequently leading to bloating or discomfort.
Yes, submerging your face in cold water triggers a primitive survival response known as the mammalian diving reflex. This sensory input immediately causes the vagus nerve to send signals that slow down your heart rate and constrict peripheral blood vessels to conserve oxygen.
5. How long does it take to see improvements in nervous system balance from a new routine?
While lifestyle adjustments like deep breathing exercises can bring a sense of physical ease within a few minutes, truly rebalancing a worn-down autonomic nervous system typically requires two to three weeks of consistent sleep, balanced nutrition, and daily movement.
⚠️ Disclaimer
This article is for educational purposes only and does not provide medical advice. If you have health concerns, always consult a qualified healthcare professional.
📚 References
- Cleveland Clinic: Clinical overviews of cranial nerve X, parasympathetic anatomy, and internal balance.
- Britannica: Comprehensive reference data tracking the physical path and organ connections of the vagus nerve.
- Johns Hopkins Medicine: Clinical reviews on autonomic regulation, heart rate variability, and medical stimulation therapies.
- Harvard Health Publishing: Research on the gut-brain connection, stress management, and lifestyle-first recovery practices.




